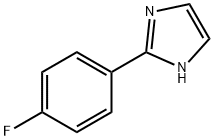
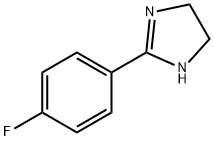
2-Aryl-4,5-dihydro-1H-imidazole (14b, x, 35 mmol) was used as the starting material and potassium carbonate (38.5 mmol) and diacetoxyiodobenzene (38.5 mmol) were added sequentially in DMSO (100 mL). The reaction mixture was placed in a dark environment and stirred overnight. Upon completion of the reaction, the reaction was quenched by the addition of water and the product was extracted with dichloromethane. The organic layers were combined, dried with magnesium sulfate and concentrated under reduced pressure to remove the solvent. The resulting residue was purified by fast column chromatography (eluent: hexane/ethyl acetate, 3:2, v/v) to afford 2-(4-fluorophenyl)-1H-imidazole as a white solid. Yield: 30%-50%.
[1] Journal of Medicinal Chemistry, 2010, vol. 53, # 20, p. 7414 - 7427
[2] Patent: WO2011/109059, 2011, A1. Location in patent: Paragraph 0040; 00354
[3] Patent: WO2014/138279, 2014, A1. Location in patent: Paragraph 00221; 00246
[4] Journal of the American Chemical Society, 2016, vol. 138, # 25, p. 7820 - 7823
[5] Heterocycles, 1995, vol. 40, # 2, p. 841 - 850