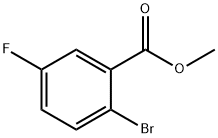
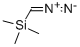Procedure for the synthesis of methyl 2-bromo-5-fluorobenzoate (159-1): trimethylsilylated diazomethane (338 mL, 676 mmol, 2.0 M ether solution) was slowly added dropwise to a stirring methanol (676 mL) solution of 2-bromo-5-fluorobenzoic acid (74 g, 338 mmol) at 0 °C until the reaction solution took on a persistent yellow color. Subsequently, acetic acid was added dropwise until the yellow color completely faded. Upon completion of the reaction, the solvent was removed by distillation under reduced pressure. The residue was dissolved in dichloromethane and filtered through a silica gel column, eluting with dichloromethane. Finally, the solvent was removed from the eluate by reduced pressure distillation to give 77 g (98% yield) of methyl 2-bromo-5-fluorobenzoate (159-1) as a yellow oil.