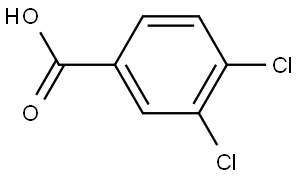
1. CDI (41.5 g, 290.32 mmol, 3.70 eq.) was added to a solution of 3,4-dichlorobenzoic acid (15 g, 78.53 mmol, 1.00 eq.) in tetrahydrofuran (200 mL) under dry conditions. The reaction mixture was stirred at 25 °C for 4 hours.
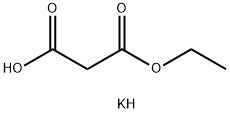
2. In another reaction flask, monoethyl malonate potassium salt (37.7 g, 235 mmol, 3.00 eq.) was dissolved in acetonitrile (400 mL), followed by the addition of MgCl2 (33.5 g, 353 mmol, 4.5 eq.) and triethylamine (23.8 g, 235 mmol, 3.00 eq.).
3. The solutions from Steps 1 and 2 were combined and the reaction was stirred at room temperature for 2 hours.
4. Upon completion of the reaction, the mixture was concentrated under reduced pressure to remove the solvent.
5. The concentrated residue was diluted with 200 mL of water and the pH was adjusted to 5 with 1 M HCl.
6. The aqueous phase was extracted with ethyl acetate (4 x 50 mL), the organic layers were combined and dried with anhydrous magnesium sulfate.
7. After filtering the desiccant, the organic phase was concentrated under reduced pressure.
8. The crude product was purified by silica gel column chromatography using petroleum ether/ethyl acetate (50:1) as eluent.
9. 15 g (73% yield) of ethyl 3-(3,4-dichlorophenyl)-3-oxopropanoate was finally obtained as a colorless oil.