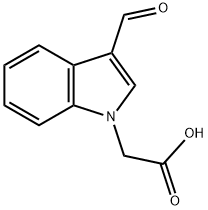
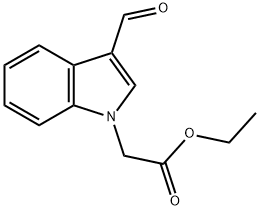
Ethyl (3-formyl-1H-indol-1-yl)acetate (15.9 g, 69 mmol) was dissolved in 1,4-dioxane (100 mL), 1 N sodium hydroxide solution (10 mL) was added, and the resulting mixture was stirred at 0 °C. The reaction mixture was then stirred at room temperature for 4 days. After completion of the reaction, water (500 mL) was added and the mixture was washed with ether (150 mL). The aqueous phase was acidified with 5 N hydrochloric acid to pH < 2 and subsequently extracted with ethyl acetate (250 mL + 150 mL) in two fractions. The organic phases were combined, dried over anhydrous magnesium sulfate, and concentrated under reduced pressure to give (3-formylindol-1-yl)acetic acid (10.3 g, 73% yield) as a white solid. The product was characterized by 1H-NMR (DMSO-d6): δH=5.20 (2H, s), 7.3 (2H, m), 7.55 (1H, d), 8.12 (1H, d), 8.30 (1H, s), 9.95 (1H, s), 13.3 (1H, br s).