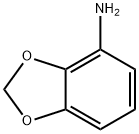
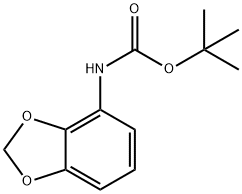
The general procedure for the synthesis of 4-amino-1,3-benzodioxole from tert-butyl benzo[d][1,3]dioxol-4-ylcarbamate was as follows: the Boc-protected feedstock G2 (257 mg, 1.08 mmol) was dissolved in a dioxane solution (5.0 mL) of 4 M HCl and stirred for 1 h at room temperature. Upon completion of the reaction, the solvent was removed by evaporation and the residue was diluted with saturated sodium bicarbonate solution (a few milliliters) and 1 M NaOH solution (1 mL), followed by extraction with ethyl acetate (EtOAc, 2×). The organic phases were combined, dried over anhydrous magnesium sulfate (MgSO), filtered and evaporated to dryness to afford the crude product 2,3-methylenedioxypropane G3 (158 mg, 106% yield).
[1] Patent: WO2004/103996, 2004, A1. Location in patent: Page 50
[2] Journal of Medicinal Chemistry, 2004, vol. 47, # 4, p. 871 - 887
[3] Journal of the Chemical Society, Perkin Transactions 1: Organic and Bio-Organic Chemistry (1972-1999), 1991, # 2, p. 259 - 262
[4] Patent: US2004/34046, 2004, A1
[5] Patent: WO2004/4732, 2004, A1. Location in patent: Page/Page column 66