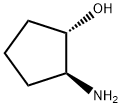
To a 50 mL autoclave was added 0.900 g (3.82 mmol) of (S,S)-2-benzyloxycarbonylaminocyclopentanol and 0.36 g of 5% palladium-carbon catalyst. Under nitrogen protection, 4.5 mL of degassed methanol was added to the reaction system. The reaction mixture was stirred under 2 MPa hydrogen pressure at 50 °C for 3 hours. Upon completion of the reaction, the palladium-carbon catalyst was removed by diatomaceous earth filtration and the filtrate was concentrated by rotary evaporator to give 0.59 g of crude product. The percentage enantiomeric excess (ee) of the compound was determined to be more than 99%.1H-NMR (200 MHz, CDCl3) δ: 1.2-1.3 (m, 1H), 1.5 (m, 1H), 1.6-1.7 (m, 2H), 1.9-2.0 (m, 2H), 2.3-2.5 (br, 3H), 3.0 (m, 1H), 3.7 ( m, 1H).
![Carbamic acid, N-[(1R,2R)-2-hydroxycyclopentyl]-, phenylmethyl ester](/CAS/20200611/GIF/672310-36-0.gif)