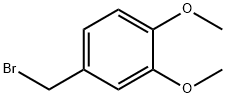
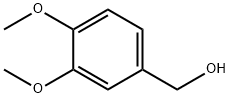
3,4-Dimethoxybenzyl Bromide is synthesized from commercially available 3,4-dimethoxybenzyl alcohol and PBr3 or
HBr.[2-3]
3,4-Dimethoxybenzyl Bromide (DMBBr) is used as a reagent for introduction of 3,4-dimethoxybenzyl protecting group, which can be cleaved selectively under
conditions of benzylic oxidation.[1]
The general procedure for the synthesis of 3,4-dimethoxybenzyl bromide from 3,4-dimethoxybenzyl alcohol: 3,4-dimethoxybenzyl alcohol (1 mmol) was dissolved in anhydrous benzene (15 mL) under anhydrous conditions, phosphorus tribromide (0.5 mL) was added and the reaction was stirred at room temperature. Upon completion of the reaction, the target product 3,4-dimethoxybenzyl bromide was obtained in quantitative yield by conventional post-treatment steps.
3,4-Dimethoxybenzyl Bromide reactions are usually carried out in polar solvents such as THF or DMF. The reagent is quite unstable and is best prepared fresh or stored at low
temperature under nitrogen. It is a lachrymator.
1. (a) Horita, K.; Yoshioka, T.; Tanaka, T.; Oikawa, Y.; Yonemitsu, O. T 1986, 42, 3021. (b) Oikawa, Y.; Tanaka, T.;
Horita, K.; Yoshioka, T.; Yonemitsu, O. TL 1984, 25, 5393.
2. Lakhlifi, T.; Sedqui, A.; Laude, B.; Dinh An, N.; Vebrel, J. CJC 1991, 69, 1156.
3. Coote, S. J.; Davies, S. G.; Middlemiss, D.; Naylor, A. JCS(P1) 1989, 2223.