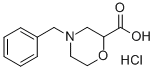
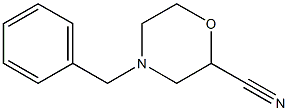
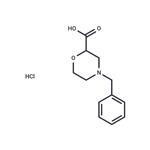
4-Benzylmorpholine-2-carbonitrile (64.1 g, 316.9 mmol) was used as the raw material, which was dissolved in 6 N aqueous hydrochloric acid (600 mL) and the reaction was heated to reflux for 2.5 hours. After the reaction was completed, the reaction solution was cooled to -10 °C. The precipitated solid product was collected by filtration and washed with acetone (300 mL) pre-cooled to -10 °C. The resulting solid was dried in an oven at 60 °C to afford 4-benzyl-2-morpholine carboxylic acid hydrochloride (78.7 g, 305.4 mmol, 96% yield). The product was characterized by 1H NMR (400 MHz, DMSO-d6): δ 2.18 (broad peak, -COOH), 7.67-7.65 (multiple peaks, 2H), 7.53-7.40 (multiple peaks, 3H), 4.61 (double peak, J = 10.8 Hz, 1H), 4.39 (multiple peaks, 2H), 4.05-4.94 (multiple peaks, 2H) , 3.43 (double peak, J = 12.0 Hz, 1H), 3.20-3.07 (multiple peaks, 3H).