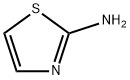
General procedure for the synthesis of tert-butyl thiazole-2-carbamate from 2-aminothiazole and di-tert-butyl dicarbonate: tert-butyl dicarbonate (27.5 ml, 119.9 mmol) was added to a tetrahydrofuran solution (20 ml) of 2-aminothiazole (10.0 g, 99.9 mmol) and the reaction mixture was stirred for 16 hours at room temperature. After completion of the reaction, the solvent was removed by distillation under reduced pressure to give the crude product. The crude product was recrystallized in hexane to give tert-butyl thiazole-2-carbamate (19.25 g, 96% yield) as colorless crystals. The melting point of the product was 181.5-182.5 °C, 1H NMR (300 MHz, CDCl3) δ 1.60 (9H, s), 6.88 (1H, d, J = 3.7 Hz), 7.38 (1H, d, J = 3.7 Hz), 11.9 (1H, s); IR (Nujol) 1713 cm-1; APCI-MS m/z 201 [M + H]+.