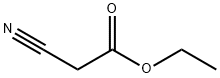
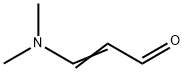
Ethyl cyanoacetate (59 mL, 0.5 mol) and 1-dodecyl-3-methylimidazolium chloride (50 mL) were added to the reactor, followed by 3-dimethylaminoacrylate (62 mL, 0.5 mol) and stirred well. The reaction mixture was heated to 90 °C in an oil bath and kept at this temperature for 4 hours. The progress of the reaction was monitored by thin layer chromatography (TLC, unfolding agent: petroleum ether/dichloromethane = 1:2, color developer: iodine vapor), and after confirming the complete reaction of 3-dimethylaminobenzaldehyde, the reaction mixture was cooled to room temperature. The reaction mixture was extracted three times with 1,2-dichloroethane (60 mL) and the organic phases were combined. The aqueous phase was washed with deionized water, dried under vacuum and recycled. Dry hydrogen bromide (HBr) gas was passed into the organic phase until completion of the reaction was confirmed by high performance liquid chromatography (HPLC). The pH of the reaction mixture was adjusted to 5-6 with 20% sodium carbonate solution and then the aqueous phase was extracted with 1,2-dichloroethane (20 mL x 3). The organic layers were combined, dried over molecular sieves, filtered and the solvent was evaporated under reduced pressure to recover 1,2-dichloroethane to give ethyl 2-bromonicotinate (108.4 g, light brown liquid) in 94.0% yield.