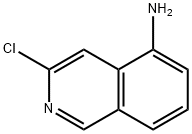
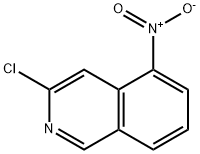
General procedure for the synthesis of 3-chloro-5-aminoisoquinoline from 3-chloro-5-nitroisoquinoline: 3-chloro-5-nitroisoquinoline (2.3 g, 11.0 mmol) was dissolved in acetic acid (100 mL), followed by the addition of zinc powder (3.5 g, 55.1 mmol) in batches. The reaction mixture was stirred at room temperature for 2 hours. After completion of the reaction, the mixture was filtered through diatomaceous earth and the filtrate was concentrated under reduced pressure. The residue was purified by silica gel column chromatography using dichloromethane solution containing 1% methanol as eluent to give the final 3-chloro-5-aminoisoquinoline as an off-white solid. The product was analyzed by mass spectrometry (ESI, m/z), which showed that the [M+1]+ peak was located at 179.0; the 1H NMR (300 MHz, DMSO-d6) δ-values were 8.98 (s, 1H), 8.13 (s, 1H), 7.40-7.35 (m, 1H), 7.28-7.25 (m, 1H), 6.90-6.87 (m, 1H) and 6.06.