The general procedure for synthesizing bisphenol A-type diethyl ether dianhydride from bisphenol A and 4-chlorophthalic anhydride is as follows: 79 g (0.63 mol) of 32 wt.% aqueous sodium hydroxide and 62.4 g (0.3 mol) of bisphenol A are added to a reactor, and stirred until completely dissolved. The reaction mixture was heated to 85°C, at which point the solution should appear clear. Subsequently, 510 g of trimethylbenzene was added as solvent and 4.2 g of benzyltriethylammonium chloride as catalyst. The reaction was started at room temperature, gradually warmed up to 172°C and continued stirring at this temperature for 15 hours. Upon completion of the reaction, the reaction mixture was cooled to 130 °C and 108 g (0.6 mol) of 4-chlorophthalic anhydride and 4.86 g of benzyltriethylammonium chloride catalyst were added. The reaction mixture was heated to 140°C to 145°C and kept at reflux for 5 hours. At the end of the reaction, it was filtered while hot and the filtrate was transferred to a crystallization kettle, cooled to room temperature and left to crystallize for 12 hours. After crystallization was completed, the precipitate was collected by filtration, washed with non-ionic water and filtered again. The precipitate was washed with 250 g of ethanol, stirred for 2 h and filtered to obtain 125 g of wet material. 120 g of bisphenol A diethyl ether dianhydride product was obtained after drying in 85% yield. The final product was in the form of white powder.
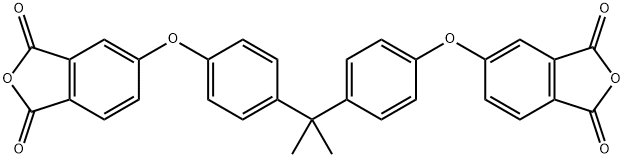
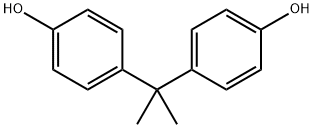
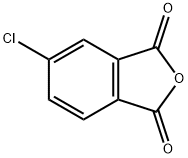

![2,2-Bis[4-(3,4-dicarboxyphenoxy) phenyl]propanedianhydride (BPADA) pictures](/ProductImageEN/2021-07/Small/ea33941e-bdd0-4ec7-8f5c-df469e11b75f.jpg)