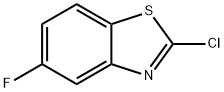
General procedure for the synthesis of 2-chloro-5-fluorobenzothiazole from 5-fluoro-2-mercaptobenzothiazole: Thionyl chloride (0.3 mL, 4.41 mmol) was slowly added dropwise to 5-fluoro-2-mercaptobenzothiazole (680 mg, 3.67 mmol). The reaction mixture was stirred at room temperature for 1 h. The reaction was then warmed up to 60 °C and continued for 40 min. Upon completion of the reaction, the reaction solution was cooled to room temperature, slowly poured into ice water and extracted with ethyl acetate (3 x 50 mL). The organic phases were combined, washed with saturated brine and dried over anhydrous sodium sulfate. The dried organic phases were concentrated under reduced pressure to afford the target product 2-chloro-5-fluorobenzothiazole (520 mg, 2.75 mmol, 75% yield).ESI-MS (M + 1): 187 (calculated value: C7H3ClFNS, 186).
[1] Patent: WO2011/143366, 2011, A1. Location in patent: Page/Page column 57
[2] Bioorganic and Medicinal Chemistry, 2015, vol. 23, # 24, p. 7661 - 7670
[3] Patent: US2004/224997, 2004, A1. Location in patent: Page 32; 33
[4] Patent: WO2017/88759, 2017, A1. Location in patent: Paragraph 00233