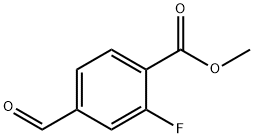
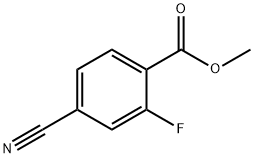
Synthesis of Example 20B Methyl 2-fluoro-4-formylbenzoate: Example 20A (methyl 4-cyano-2-fluorobenzoate, 310 mg, 1.73 mmol) was dissolved in 60% aqueous acetic acid (10 mL) and heated until completely dissolved. Nickel Ruanne (60 mg) was then added and the reaction was stirred at room temperature under hydrogen atmosphere for 18 hours. Upon completion of the reaction, the solid catalyst was removed by filtration and the filtrate was concentrated under reduced pressure. The resulting crude product was purified by silica gel column chromatography with the eluent being a 1:4 (v/v) ethyl acetate/hexane solvent mixture, resulting in the target product methyl 2-fluoro-4-formylbenzoate (220 mg, 70% yield). Mass spectrometry (DCI) analysis showed m/z 183 (M + H)+.
[1] Patent: US2007/112047, 2007, A1. Location in patent: Page/Page column 16
[2] Patent: US2007/259937, 2007, A1. Location in patent: Page/Page column 13
[3] Patent: WO2015/25164, 2015, A1
[4] Patent: US2015/57298, 2015, A1