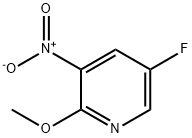
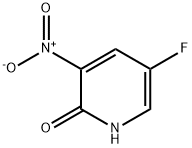
Method xxi - Pyridinylamine 1: 5-fluoro-3-nitropyridin-2-ol (158 mg, 1 mmol) was dissolved in n-hexane (1 mL), followed by the addition of silver carbonate (331 mg, 1.2 mmol) and iodomethane (0.2 mL, 2 mmol). The reaction mixture was placed in a microwave reactor and stirred at 150 °C for 1 h at 250 W power. After completion of the reaction, the solvent was removed by rotary evaporation. The residue was dissolved in ethyl acetate (2 mL) and washed twice with water (2 mL each). The solvent was again removed from the organic phase by rotary evaporation, and the crude product obtained was purified using an ISCO automated fast chromatography system to give 5-fluoro-2-methoxy-3-nitropyridine (100 mg, 58% yield) as a colorless oil. The structure of the product was confirmed by 1H NMR (400 MHz, CDCl3): δ 8.31 (d, J = 2.8 Hz, 1H), 8.09 (dd, J = 7.2, 2.8 Hz, 1H), 4.11 (s, 3H).