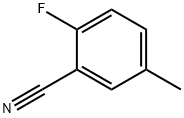
General procedure for the synthesis of 2-fluoro-5-(bromomethyl)benzonitrile from 2-fluoro-5-methylbenzonitrile: 2-fluoro-5-methylbenzonitrile (3.00 g, 22 mmol), N-bromosuccinimide (4.12 g, 23 mmol), and benzoyl peroxide (1.07 g, 4.4 mmol) were dissolved in carbon tetrachloride (100 mL) and heated to reflux for 6 hours. Upon completion of the reaction, the mixture was cooled to room temperature and subsequently mixed with water. The organic phase was separated, dried with anhydrous magnesium sulfate and filtered through diatomaceous earth (Celite). The filtrate was concentrated under reduced pressure and purified by fast column chromatography (n-hexane/ethyl acetate, 20:1) to afford 5-(bromomethyl)-2-fluorobenzonitrile (1.58 g, 33% yield) as an oil.1H NMR (CDCl3, 400 MHz) data were as follows: δ 4.45 (s, 2H), 7.21 (m, 1H), 7.65 (m, 2H).
[1] Journal of Medicinal Chemistry, 2016, vol. 59, # 20, p. 9422 - 9430
[2] Bioorganic and Medicinal Chemistry Letters, 2002, vol. 12, # 20, p. 2925 - 2930
[3] Helvetica Chimica Acta, 2017, vol. 100, # 12,
[4] Patent: WO2010/93845, 2010, A1. Location in patent: Page/Page column 86
[5] Bioorganic and Medicinal Chemistry Letters, 1998, vol. 8, # 7, p. 715 - 720