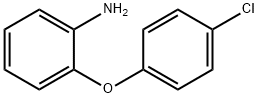
General procedure for the synthesis of 4'-chloro-2-amino-diphenyl ether from 1-(4-chlorophenoxy)-2-nitrobenzene: A mixture of commercially available iron powder (3.18 g, 57.28 mmol) and ammonium chloride (383 mg, 7.15 mmol) in ethanol (45 mL) and water (8 mL) was refluxed. To this mixture was added 1-(4-chlorophenoxy)-2-nitrobenzene (AMR01028,2.2 g, 8.81 mmol) and the resulting mixture was stirred at reflux for 1 hour. After completion of the reaction, the solvent was removed and the residue was diluted in aqueous sodium bicarbonate (40 mL) and extracted with dichloromethane (3 x 20 mL). The organic layers were combined, dried with anhydrous magnesium sulfate, filtered and the solvent was evaporated to give 4'-chloro-2-amino-diphenyl ether (AMR01029,1.50 g, 78%) as a colorless oil, which could be used in the next step without further purification. Thin layer chromatography (TLC) Rf value was 0.53 (unfolding agent: dichloromethane).1H NMR (270MHz, CDCl3) data: δ 3.77 (2H, broad single peak, NH2), 6.71 (1H, multiple peaks), 6.83 (2H, multiple peaks), 6.89 (2H, AA'BB' system), 6.99 (1H, multiple peaks), 7.25 ( 2H, AA'BB' system).
![1-[(4-Chlorophenyl)oxy]-2-nitrobenzene](/CAS/GIF/39145-47-6.gif)