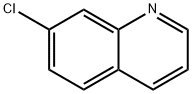
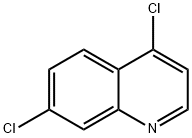
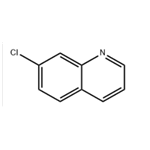
General Program 138: Synthesis of 7-chloroquinoline (Intermediate 573)
1. 4,7-Dichloroquinoline (10 g, 50 mmol) was dissolved in THF (100 ml) and degassed using N2 for 5 min.
2. PdCl2 (dppf) (1.2 g, 2 mmol), TMEDA (9.97 g, 86 mmol) and NaBH4 (3.24 g, 86 mmol) were added sequentially.
3. The reaction mixture was stirred at room temperature for 5 hours.
4. Upon completion of the reaction, brine (20 ml) was added dropwise followed by removal of solvent under vacuum.
5. The residue was dissolved in EtOAc (200 ml), dried by adding MgSO4 and then concentrated under vacuum.
6. The crude product was purified by column chromatography using heptane/EtOAc (4:1 to 1:1 gradient) as eluent to afford the title compound 7-chloroquinoline (5.4 g, 65% yield).
MW: 163.61
HPLCMS (Method B): [m/z]: 163.90