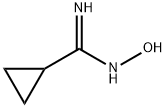
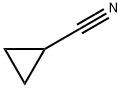
General procedure for the synthesis of N-hydroxycyclopropanecarboxamidine from cyclopropanecarbonitrile: A mixture of cyclopropanecarbonitrile (5 g, 74.6 mmol) and hydroxylamine (50 wt% aqueous, 5.91 g, 89.5 mmol) in ethanol (15 mL) was reacted with stirring at 80 °C overnight. Upon completion of the reaction, the mixture was cooled to room temperature and the volatile solvent was subsequently removed under reduced pressure to afford the crude product N-hydroxycyclopropanecarboxamidine (7.0 g, 94% yield). The product could be directly used in the subsequent reaction without further purification. The product was analyzed by LCMS showing m/z = 101.2 [M + H]+, which was consistent with the theoretically calculated value (exact mass of C4H8N2O: 100.1).1H NMR (400 MHz, DMSO-d6) data were as follows: δ 0.54-0.60 (m, 2H), 0.61-0.66 (m, 2H), 1.28-1.36 (m, 1H) , 5.18 (bs, 2H), 8.68 (bs, 1H).