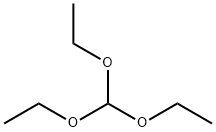
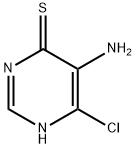
General procedure for the synthesis of 7-chlorothiazolo[5,4-d]pyrimidine from triethyl orthoformate and 5-amino-6-chloropyrimidine-4-thiol: 5-amino-6-chloropyrimidine-4-thiol (4 g, 24.75 mmol) was dissolved in triethyl orthoformate and heated and refluxed for 1 hour. Upon completion of the reaction, the reaction mixture was concentrated to 60% of the original volume and subsequently cooled in a refrigerator. The precipitated solid was collected by filtration and dried under vacuum to afford 7-chlorothiazolo[5,4-d]pyrimidine (2.8 g, 66.04% yield) as a brown solid.1H NMR (300 MHz, CDCl3): δ 9.22 (s, 1H), 8.97 (s, 1H).
[1] Patent: WO2010/14930, 2010, A2. Location in patent: Page/Page column 50
[2] Patent: WO2008/57402, 2008, A2. Location in patent: Page/Page column 61-62
[3] Chemical and pharmaceutical bulletin, 1968, vol. 16, # 4, p. 750 - 755
[4] Patent: US2012/122838, 2012, A1. Location in patent: Page/Page column 81
[5] Patent: WO2013/12918, 2013, A1. Location in patent: Paragraph 00533
![7-chlorothiazolo[5,4-d]pyrimidine Structure](/CAS/GIF/13316-12-6.gif)
![7-Chlorothiazolo[5,4-d]pyriMidine pictures](https://img.chemicalbook.com/ProductImageEN/2020-5/Small/94428527-44cf-4fc0-9bea-b9c33ca4c8a5.jpg)