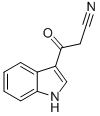
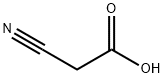
GENERAL METHOD: Cyanoacetic acid (0.363 g, 4.26 mmol) was dissolved in acetic anhydride (8 mL), stirred and heated to 50 °C until the solid was completely dissolved. Indole (0.5 g, 4.26 mmol) was then added and the reaction mixture was heated to 85°C and maintained for 5 minutes. Upon completion of the reaction, the mixture was cooled to 0 °C and the precipitate was collected by diafiltration and washed with ice-cold methanol (2 x 5 mL) to afford 3-(1H-indol-3-yl)-3-oxopropanenitrile in 76% yield. Next, an ethanol solution (10 mL) of 3-oxo-3-(1H-indol-3-yl)propionitrile (1.08 mmol) was mixed with an ethanol solution (10 mL) of 4-chlorobenzaldehyde (1.14 mmol) and heated to 70 °C. Piperidine (2 drops) was then added and heating was continued to reflux for 2 hours. At the end of the reaction, the mixture was cooled and the solvent was removed under reduced pressure to give a yellow solid. The crude product was purified by ethanol recrystallization to give the target compound (14) as a yellow solid in 80% yield with a melting point of 230-232 °C.
[1] Journal of Heterocyclic Chemistry, 2007, vol. 44, # 1, p. 109 - 114
[2] Indian Journal of Chemistry - Section B Organic and Medicinal Chemistry, 2013, vol. 52, # 6, p. 810 - 813
[3] Synthesis, 2004, # 16, p. 2760 - 2765
[4] Patent: WO2008/87529, 2008, A1. Location in patent: Page/Page column 126
[5] Patent: CN107698565, 2018, A. Location in patent: Paragraph 0037; 0038