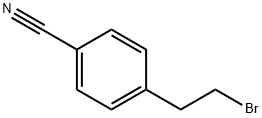
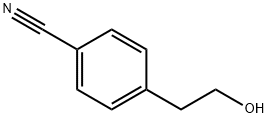
General procedure for the synthesis of 4-(2-bromoethyl)benzonitrile from 4-(2-hydroxyethyl)benzonitrile: to a stirred solution of 4-(2-hydroxyethyl)benzonitrile (1.0 g, 6.8 mmol, 1.0 eq.) in dichloromethane (DCM, 20 mL) was added sequentially carbon tetrabromide (CBr4, 4.5016 g, 13.6 mmol, 2.0 eq.) and triphenylphosphine (TPP, 3.835 g, 13.6 mmol, 2.0 equiv). The reaction mixture was stirred at room temperature for 5 hours and the progress of the reaction was monitored by thin layer chromatography (TLC). Upon completion of the reaction, the reaction mixture was diluted with water and extracted with ethyl acetate (EtOAc). The organic layers were combined, dried with anhydrous sodium sulfate, filtered and the filtrate was concentrated. The crude product was purified by column chromatography to afford pure 4-(2-bromoethyl)benzonitrile (1.3 g, 91% yield). The results of liquid chromatography-mass spectrometry (LC-MS, Method 23): retention time (Rt) = 2.01 min; mass-to-charge ratio (m/z) = 210.05 ([M+H]+).
[1] Patent: WO2018/149986, 2018, A1. Location in patent: Page/Page column 95
[2] Pharmazie, 1982, vol. 37, # 1, p. 13 - 16
[3] ACS Medicinal Chemistry Letters, 2014, vol. 5, # 1, p. 65 - 68
[4] Journal of the Chemical Society, Perkin Transactions 2: Physical Organic Chemistry (1972-1999), 1979, p. 1128 - 1132
[5] Patent: US5811459, 1998, A