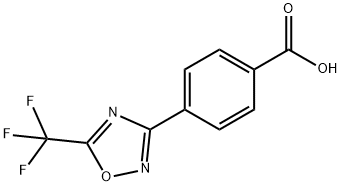
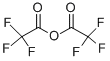
General procedure for the synthesis of 4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzoic acid from trifluoroacetic anhydride and 4-[(Z)-amino(hydroxyiminomethyl)methyl]benzoic acid: a solution of tetrahydrofuran (2.5 L) of 4-[(Z)-N'-hydroxyformamidinyl]benzoic acid (200 g, 1.0 equiv) was treated with trifluoroacetic anhydride (350 g, 1.5 equivalent) treatment. The reaction mixture was stirred at room temperature overnight. After completion of the reaction, the reaction mixture was diluted with methyl tert-butyl ether and washed with saturated aqueous sodium bicarbonate. The organic layer was separated and concentrated under reduced pressure to remove the solvent to give the crude product. The crude product was purified by recrystallization from diisopropyl ether to afford 4-[5-(trifluoromethyl)-1,2,4-oxadiazol-3-yl]benzoic acid as a light brown solid (220 g, 76% yield).1H NMR (400 MHz, DMSO-d6, 298 K): δ [ppm] = 13.40 (broad single peak, 1H), 8.22-8.10 (multiple peaks 4H).
![4-[(Z)-Amino(hydroxyimino)methyl]benzoic acid](/CAS/20140525/GIF/CB82556967.gif)
