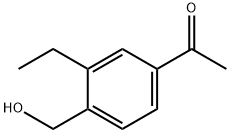
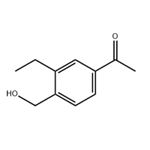
1-Iodo-4-bromo-2-ethylbenzene was synthesized from 2-ethylaniline by a one-pot reaction of aminoprotection, bromination, deprotection and iodination, and then 4-bromo-2-ethylbenzyl alcohol was synthesized from 4-bromo-2-ethylbenzyl alcohol by a one-pot reaction of Gergel exchange aldolization and reduction, and 1-(3-ethyl-4-hydroxymethyl) was synthesized from 1-(3-ethyl-4-hydroxymethyl) phenyl ketone by a total reaction of hydroxylation, Gergel reaction, acetylation and deprotection in 9 steps. The total yield of the reaction was 50.9%, and the GC content of the product was 98.3%.