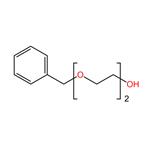
Benzyl-PEG3-alcohol is a PEG linker with an acid labile, benzyl protecting group and a reactive primary alcohol. The primary alcohol can react and further derivatize the compound. The hydrophilic PEG linker increases the water solubility of the compound in aqueous media.
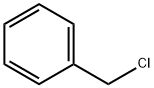
GENERAL METHOD: Dioxane (15 mL), benzyl chloride (25 mmol) and tetrabutylammonium bromide (0.4 g, 1.2 mmol) were sequentially added to a reaction flask, followed by addition of the mixture to a solution of diethylene glycol (75-125 mmol) containing KOH (1.6 g, 27.5 mmol). The reaction system was heated to 100-105 °C and the reaction was continuously stirred for 5-10 hours. Upon completion of the reaction, the reaction mixture was diluted with CHCl3 (15 mL) and water (10 mL). The organic layer was separated, washed with water (3 x 20 mL) and dried over anhydrous Na2SO4. The organic phase was concentrated under reduced pressure and the residue was purified by distillation through a Vigreux column to afford the target product 2-[2-(phenylmethoxy)ethoxy]ethanol. The specific reaction conditions, yields, physicochemical parameters and 1H NMR data are detailed in Table 6 of the literature.
[1] Synthetic Communications, 1986, vol. 16, # 1, p. 19 - 26
[2] Carbohydrate Research, 1986, vol. 150, p. 199 - 212
[3] Russian Chemical Bulletin, 2014, vol. 63, # 11, p. 2445 - 2454
[4] Izv. Akad. Nauk, Ser. Khim., 2014, # 11, p. 2445 - 2454
[5] Analytical Chemistry, 1992, vol. 64, # 15, p. 1685 - 1690
![2-[2-(BENZYLOXY)ETHOXY]ETHANOL Structure](/CAS/GIF/2050-25-1.gif)