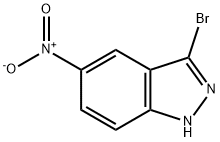
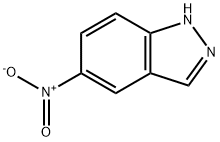
Example 22 Synthesis of 3-bromo-5-nitroindazole: 5-nitroindazole (10 g, 61.3 mmol) was dissolved in acetic acid (170 mL) and heated to 80 °C. Bromine (3.1 mL, 60.7 mmol) was added slowly and dropwise. The reaction mixture was heated to reflux. After 2 hours of reaction, it was cooled to room temperature and the precipitate precipitated was collected by filtration. The filtrate was concentrated under reduced pressure to separate the additional product and the concentrated residue was partitioned between chloroform and saturated sodium bicarbonate solution, the organic phase was separated and dried over anhydrous sodium sulfate. Decompression concentration of the organic phase afforded a solid, which was combined with the aforementioned precipitate to afford the target compound 3-bromo-5-nitroindazole as a yellow solid (11.4 g, 77% yield).1H NMR (DMSO-d6) δ: 7.74 (1H, d, J=8.0 Hz), 8.21 (1H, dd, J=8.0, 1.5 Hz), 8.40 (1H, d, J=1.5 Hz), 14.5 Hz). 1.5 Hz), 14.06 (1H, brs); MS (ES-) m/z: 240 [M-H]-.