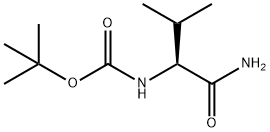
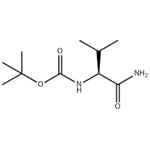
General procedure for the synthesis of tert-butyl (S)-(1-amino-3-methyl-1-oxo-2-butyl)carbamate from Boc-L-valine: to a colorless solution of 150 mg (0.50 mmol) of Boc-L-valine in 10 mL of THF was added 67 μL (0.70 mmol, 1.4 eq.) of ethyl chloroformate at 0 °C and 209 μL ( 1.5 mmol, 3.0 eq.) triethylamine. After stirring at 0 °C for 30 min, 0.75 mL of 1.0 M aqueous ammonium chloride (0.75 mmol, 1.5 equiv) was added to the colorless suspension. The mixture was continued to be stirred at 0 °C for 30 min, followed by the addition of 5 mL of water. The colorless clear solution was extracted with 30 mL of ethyl acetate and the aqueous layer was extracted with another 20 mL of ethyl acetate. The organic layers were combined, washed with 5 mL of brine and dried with anhydrous magnesium sulfate. The crude product was purified by silica gel column chromatography (eluent: ethyl acetate) to give 129 mg (86% yield) of tert-butyl (S)-(1-amino-3-methyl-1-oxo-2-butyl)carbamate. The product was a colorless solid, melting point: 149-152 °C; [α]30D = -2.4 (c 1.00, methanol); 1H NMR (400 MHz, CDCl3): δ 0.94 (d, J = 6.8 Hz, 3H, CH3CH), 0.99 (d, J = 6.8 Hz, 3H, CH3CH), 1.45 (s, 9H, (CH3)3C ), 2.16 (ddd, J = 6.7,6.8,6.8 Hz, 1H, CH(CH3)2), 3.96 (dd, J = 6.7,7.8 Hz, 1H, CHCO), 5.03,5.42,5.89 (br, br, br, 1H, 1H, 1H, NH, NH2); 13C NMR (100 MHz, CDCl3) : δ17.8,19.3,28.3,30.8,59.5,79.9,156.0,174.4; IR (KBr, vmax/cm-1): 3386 (CONH), 3345 (CONH), 3205 (CONH), 1680 (CON), 1641 (CON); HRMS (ESI-TOF): Calculated value for C10H20N2O3Na (M + Na)+: 239.1366, measured value: 239.1340. Enantiomeric ratio was determined by HPLC (Chiralcel OD column: hexane/2-propanol = 95/5): retention time 9.4 min.