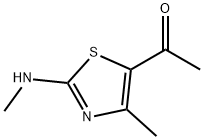
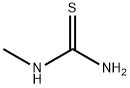
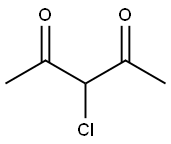
General procedure for the synthesis of 1-(4-methyl-2-(methylamino)thiazol-5-yl)ethanone from N-methylthiourea and 3-chloro-2,4-pentanedione: 1-methylthiourea (6.00 g, 66.6 mmol) was dissolved in a mixture of methanol (130 mL) and pyridine (5 mL) and cooled in an ice bath. Subsequently, 3-chloropentane-2,4-dione (8.96 mL, 66.6 mmol) was added slowly and dropwise to this cooled solution. The reaction mixture was gradually warmed to room temperature and stirred continuously for 4 hours. Upon completion of the reaction, the resulting white solid precipitate was collected by filtration and washed with ether (20 mL) to give the final target product 1-(4-methyl-2-(methylamino)thiazol-5-yl)ethanone (8.94 g, 79% yield). The product was characterized by 1H-NMR (DMSO-d6): δ 2.39 (s, 3H, CH3), 2.49 (s, 3H, CH3), 2.91 (s, 3H, CH3), 9.12 (s, 1H, NH). Mass spectrometry (ESI) analysis showed a calculated value of 171.06 (C7H11N2OS+) for m/z [M+H]+ and a measured value of 171.06.
[1] European Journal of Medicinal Chemistry, 2017, vol. 139, p. 762 - 772
[2] Journal of Medicinal Chemistry, 2004, vol. 47, # 7, p. 1662 - 1675
[3] Journal of Medicinal Chemistry, 2010, vol. 53, # 11, p. 4367 - 4378
[4] Bioorganic and Medicinal Chemistry, 2011, vol. 19, # 10, p. 3135 - 3140
[5] Bioorganic and Medicinal Chemistry, 2011, vol. 19, # 24, p. 7349 - 7356