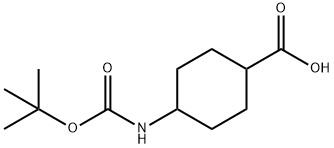
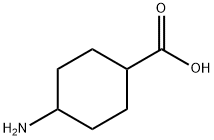
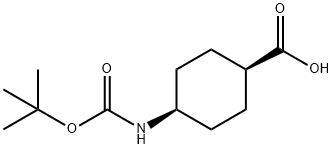
To a stirred solution of trans-4-aminocyclohexanecarboxylic acid (1 g, 6.98 mmol) in tert-butanol (10 mL) and NaOH (0.307 g, 7.68 mmol) in water (10 mL) was slowly added di-tert-butyl dicarbonate (1.7 g, 7.68 mmol) at 0 °C. The reaction mixture was stirred at room temperature overnight. Upon completion of the reaction, hexane (50 mL) was added to the mixture and the pH was adjusted to about 6 with 6 N HCl. Subsequently, the mixture was extracted with ethyl acetate (3 x 50 mL) and the organic phases were combined and washed with saturated brine solution (25 mL). The organic phase was dried over anhydrous sodium sulfate and concentrated under reduced pressure to give the white powdery product cis-4-((tert-butoxycarbonyl)amino)cyclohexanecarboxylic acid (1.4 g, 82.8% yield). Mass spectrum (ES-) m/z 242 [M-H]-.
[1] Patent: US2017/348315, 2017, A1. Location in patent: Paragraph 0400
[2] Patent: WO2010/45987, 2010, A1. Location in patent: Page/Page column 43-44
[3] European Journal of Medicinal Chemistry, 2001, vol. 36, # 3, p. 265 - 286
[4] ACS Medicinal Chemistry Letters, 2018, vol. 9, # 2, p. 89 - 93