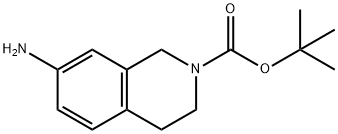
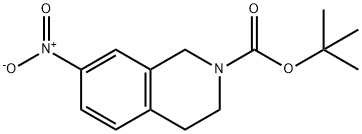
A mixed solution of 7-nitro-3,4-dihydroisoquinoline-2(1H)-carboxylic acid tert-butyl ester (33.2) (4.4 g, 15.81 mmol) was stirred with a mixed solution of Pd/C (500 mg, 4.69 mmol) in MeOH (100 mL) for 3 h at room temperature in an H2 atmosphere. Upon completion of the reaction, the reaction mixture was filtered to remove the catalyst. The filtrate was concentrated to afford the target product tert-butyl 7-amino-3,4-dihydroisoquinoline-2(1H)-carboxylate (33.3) as a yellow liquid (3.8 g, 96.8% yield). The product was characterized by 1H NMR (400 MHz, CDCl3): δ 1.48 (s, 9H), 2.71 (t, 2H), 3.58 (s, 4H), 4.47 (s, 2H), 6.44 (s, 1H), 6.52 (dd, 1H), 6.91 (d, 1H).