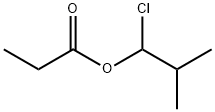
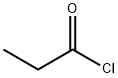
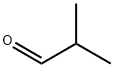
Isobutyraldehyde (3 g, 41.6 mmol) was slowly added dropwise to propionyl chloride (3.85 g, 41.6 mmol) cooled to 0-5 °C under nitrogen protection, keeping the temperature in the range 0-5 °C. The reaction mixture was stirred continuously at this temperature for 4 hours. Upon completion of the reaction, the mixture was vacuum distilled at 50 °C to remove the volatile components, resulting in 2.59 g of a colorless liquid product, i.e., isobutyl propionyl-1-chloride (yield: 38%). The structure of the product was confirmed by 1H-NMR and 13C-NMR: 1H-NMR (CDCl3, 500MHz) δ 1.06 (t, J = 7.0Hz, 6H), 7.60 (t, J = 7.5Hz, 3H), 2.13-2.17 (m, 1H), 2.39 (q, J = 7.5Hz, 2H), 6.31 (d, J = 4.5 Hz, 1H); 13C-NMR (CDCl3, 125MHz) δ 8.66, 17.25, 17.36, 27.49, 35.19, 88.61, 172.16.