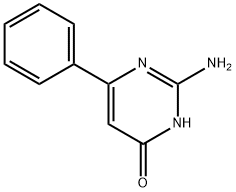
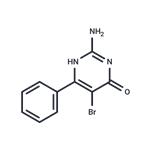
General procedure for the synthesis of 2-amino-5-bromo-4-hydroxy-6-phenylpyrimidines from 2-amino-4-hydroxy-6-phenylpyrimidines: to a suspension of 2-amino-6-phenylpyrimidin-4(3H)-one (200 mg, 1.07 mmol) in glacial acetic acid (2 mL) was added slowly and dropwise at 70 °C with bromine (57.0 μL, 1.12 mmol) in a glacial acetic acid (2 mL) solution. The reaction mixture was cooled to room temperature and stirring was continued for 4 hours. Upon completion of the reaction, the solvent was removed by distillation under reduced pressure. The resulting residue was ground with ether (10 mL) and recrystallized from ethanol to give 2-amino-5-bromo-4-hydroxy-6-phenylpyrimidine as a colorless solid (171 mg, 60% yield). The melting point of the product was 275-277 °C (literature value: 285-287 °C); high resolution mass spectrometry (HRMS) measured value: M+H+, 264.9847 (C10H9 79BrN3O+, calculated value 264.9845); infrared spectra (νmax, CHCl3, cm-1): 3691, 3607, 3012, 1692, 1602 , 1473, 1440, 1328, 1240; NMR hydrogen spectrum (1H NMR, 400 MHz, DMSO-d6): δ 7.56-7.53 (2H, m, ArH), 7.45-7.43 (3H, m, ArH), 6.87 (2H, br s, NH2); NMR carbon spectrum (13C NMR, 100 MHz. DMSO-d6): δ 158.6 (C), 153.2 (2×C), 136.3 (C), 129.7 (CH), 128.5 (CH), 128.0 (CH), 97.3 (C); mass spectrum (ESI): m/z 264/266 (M+H+, 34/35%). The spectral data of the obtained product are in agreement with literature reports.