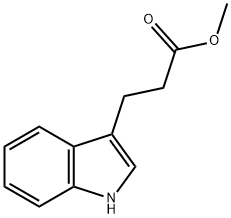
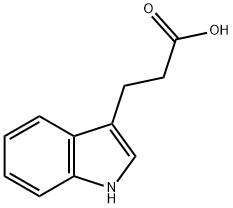
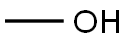(a) Synthesis of methyl 3-(1H-indol-3-yl)propionate: 3-indolepropionic acid (5 g, 26.4 mmol) was dissolved in methanol (80 mL) under stirring conditions, followed by the addition of concentrated hydrochloric acid and sulfuric acid (0.5 mL). The reaction mixture was heated to reflux for 5 hours. Upon completion of the reaction, the mixture was cooled to room temperature and concentrated to 1/3 of the original volume under reduced pressure. the remaining solution was diluted with water and extracted with dichloromethane. The organic phase was washed sequentially with saturated aqueous sodium bicarbonate and brine, followed by drying with anhydrous sodium sulfate. The crude product was purified by silica gel column chromatography with the eluent ethyl acetate-hexane (0 to 30% gradient), resulting in 5.16 g (96% yield) of the target compound as a light yellow oil, which solidified on standing.
[1] Tetrahedron Letters, 2009, vol. 50, # 50, p. 7035 - 7037
[2] Bioorganic and Medicinal Chemistry, 2006, vol. 14, # 14, p. 4958 - 4979
[3] Patent: WO2010/132615, 2010, A1. Location in patent: Page/Page column 100-101
[4] Bioorganic and Medicinal Chemistry Letters, 2009, vol. 19, # 24, p. 6926 - 6930
[5] Patent: WO2011/56630, 2011, A2. Location in patent: Page/Page column 64-65