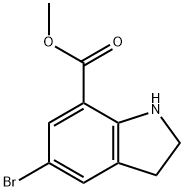
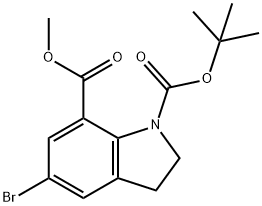
General procedure for the synthesis of methyl 5-bromodihydroindole-7-carboxylate from methyl 1-BOC-5-bromoindoline-7-carboxylate: 1-(1,1-dimethylethyl)-7-methyl-5-bromo-2,3-dihydro-1H-indole-1,7-dicarboxylate (9 g, 25 mmol) was dissolved in trifluoroacetic acid (6 mL). The reaction mixture was stirred at room temperature for 16 hours. Upon completion of the reaction, dichloromethane and 2 M sodium hydroxide solution were added and the organic layer was washed twice with sodium hydroxide solution until the aqueous layer was pH > 7. Subsequently, the organic layer was concentrated under vacuum to afford 6.5 g (100% yield) of methyl 5-bromodihydroindole-7-carboxylate as a brown solid. The product was characterized by 1H NMR (400 MHz, DMSO-D6): δ 2.99 (t, J=8.5 Hz, 2H), 3.61 (t, J=8.4 Hz, 2H), 3.78 (s, 3H), 6.72 (s, 1H), 7.28 (d, J=1 Hz, 1H), 7.46 (d, J=2 Hz, 1H); mass spectrometry analysis showed m/ z 256/258 (1:1 ratio) (M+1)+ with a retention time of 3.32 min.