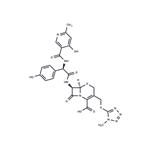
Yellow crystal, melting point 213~215°C (decomposition). Cefpiramide Sodium (Cefpiramide Sodium): C25H23N8O7S2Na.[74849-93-7]. White to yellowish white crystalline powder, odorless, bitter taste. Very soluble in dimethylformamide, soluble in water, slightly soluble in methanol, very slightly soluble in ethanol, insoluble in chloroform, acetone, tetrahydrofuran, acetonitrile or n-hexane.
Cefpiramide is used as a β-lactam antibiotic.
ChEBI: A third-generation cephalosporin antibiotic with [(1-methyl-1H-tetrazol-5-yl)sulfanyl]methyl and (R)-2-{[(4-hydroxy-6-methylpyridin-3-yl)carbonyl]amino}-2-(4-hydroxyphenyl)acetamido groups at positions 3 and 7, respectiv
ly, of the cephem skeleton. It has a broad spectrum of antibacterial activity.
A semisynthetic parenteral cephalosporin. It exhibits a broad
range of activity, which includes Ps. aeruginosa, though the overall activity is rather modest. It is moderately
stable to most β-lactamases but less so than ceftazidime or
cefpirome.
In volunteers given 0.5 or 1 g by intravenous bolus, the mean
plasma concentration shortly after injection was around 150 or
300 mg/L, respectively. There was no accumulation when the
same doses were repeated every 12 h for 11 doses. It is highly
bound to plasma protein (c. 95%). The mean plasma half-life
is around 5 h. Less than one-quarter of the dose appears in the
urine over 24 h; the rest is excreted in bile and high concentrations
are found in feces. Renal impairment has little effect on
elimination in patients with normal liver function.
Diarrhea may be associated with marked suppression of
gut flora resulting from biliary excretion of the drug. The molecule
includes a C-3 methylthiotetrazole side chain and side
effects associated with that substituent are to be expected.
It is available in Japan.
(1) Sodium compound D-a-(4-hydroxy-6-methylnicotinamide)-P-hydroxyphenylacetate was dissolved in an organic solvent and activated by an activator catalyzed by a base at a temperature of -25 to -20C to produce a solution of mixed anhydride (a);
(2) the compound 7-amino-3-(1 -methyl-1H-tetrazole-5-thiomethyl)-8-oxo-5 -thia-1-azabicyclo[4.2.0]oct-2-ene-2-carboxylic acid (7-TMCA ) was dissolved in an organic solvent and a silanization reagent was added at a temperature of 20-25C to carry out the silylation protection reaction, and after the reaction was complete, it was cooled down to -25~-20C to obtain solution (b);
(3) solution (a) was mixed with solution (b), and to the resulting mixed solution Triethylamine was added, and after the reaction was complete at a temperature of -25~-20C, it was added to an aqueous solution and the pH was adjusted to obtain cefpiramide.
Cefpiramide (25 mg/kg; i.v.; once) shows anti-Streptococcus pneumoniae activity in experimental pneumococcal meningitis[2].
Cefpiramide (25 mg/kg; i.v.; once) reduces bacteria in CSF more than 10[4] CFU/ml[2].
| Animal Model: | New Zealand white male rabbits(2-3 kg)[2]. |
| Dosage: | 25 mg/kg |
| Administration: | Intravenous injection; once. |
| Result: | Anti-Streptococcus pneumoniae. |