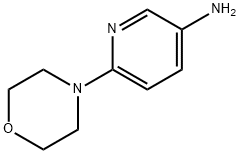
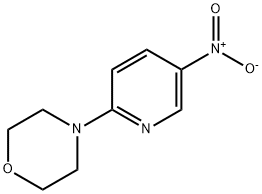
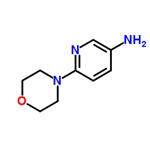
General procedure for the synthesis of 3-amino-6-morpholinopyridine from 4-(5-nitropyridin-2-yl)morpholine: a mixture of 1.65 g (7.8 mmol) of 4-(5-nitro-2-pyridinyl)morpholine, 160 mg of 5% Pt/C catalyst, and 20 mL of ethanol was placed in a reactor. The reaction was carried out under 50 psi hydrogen atmosphere for 5 hours. Upon completion of the reaction, the mixture was filtered through a pad of diatomaceous earth to remove the catalyst, followed by evaporation of the solvent under reduced pressure to afford 1.4 g (100% yield) of 6-(4-morpholino)-3-pyridinamine, the product being a purple solid. Its 1H NMR (400 MHz, DMSO-d6) data were as follows: δ 7.60 (d, J=2.9 Hz, 1H), 6.92 (dd, J=8.8 and 2.9 Hz, 1H), 6.62 (d, J=8.8 Hz, 1H), 4.59 (brs, 2H), 3.65-3.72 (m, 4H), 3.17 (dt, J= 4.9 and 2.4 Hz, 4H).