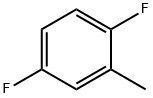
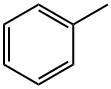
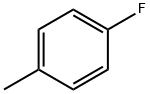
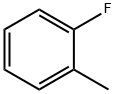
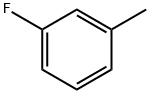
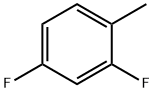
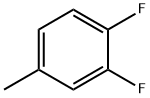
General procedure for the synthesis of p-fluorotoluene, 2-fluorotoluene, 3-fluorotoluene, 2,4-difluorotoluene, 2,5-difluorotoluene, and 1,2-difluoro-4-methylbenzene from toluene: To a glass reactor fitted with a PTFE-lined magnetic stirring bar connected to a gas scrubbing flask, add toluene (0.95-1.14 mmol), acetonitrile (1-2.5 mL/mmol toluene ) and boron trifluoride ethyl ether complex (1.3-1.6 mmol/mmol toluene). The reaction solution was stirred at -25 °C for 10-15 min, followed by the addition of xenon difluoride (1.2-1.3 mmol/mmol toluene) in batches. The reaction mixture was slowly warmed to 22 °C over 1 h and stirring was continued for 40-60 min. Upon completion of the reaction, hexafluorobenzene was added as an internal standard and samples were taken for 19F NMR analysis (Method B). The reaction mixture was quenched with saturated aqueous sodium bicarbonate and extracted with dichloromethane (1-2 mL). The organic phases were combined, dried with anhydrous magnesium sulfate and subsequently analyzed by 19F NMR and GC/MS (Procedure C). The major products are listed in the table and the remaining products are listed below (GC/MS data).