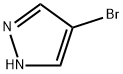
White to cream crystalline powder
As a mono substituted pyrozole, 4-Bromopyrazole can be used as Inhibitor of human liver alcohol dehydrogenase and used in mutagenicity study with the L-arabinose resistance test of Salmonella typhimurium.
4-Bromopyrazole can be used as starting material employed in a synthesis of 1,4′-bipyrazoles and in suzuki reaction.
4-Bromopyrazole is a pyrazole derivative. It is reported to react with titanium tetrachloride to afford binary adducts. Mutagenicity of 4-bromopyrazole has been tested using the L-arabinose forward mutation assay of Salmonella typhimurium. It is reported to inhibit the oxidative phosphorylation, the ATP-32P exchange reaction, and energy dependent and independent calcium uptake.
Step 1: 1H-pyrazole (10 g, 147 mmol) was suspended in water (150 mL) at room temperature and N-bromosuccinimide (NBS, 26.1 g, 147 mmol) was added all at once. The reaction mixture immediately turned milky white, followed by continuous stirring at room temperature for 24 hours. Upon completion of the reaction, the reaction mixture was extracted with ethyl acetate (EtOAc, 2 x 100 mL). The combined organic phases were washed sequentially with aqueous sodium carbonate (Na2CO3) and saturated saline and dried over anhydrous sodium sulfate (Na2SO4). After concentration under reduced pressure, 21.5 g of light brown oily product (100% yield) was obtained, which solidified to a solid after standing. High performance liquid chromatography (HPLC) analysis showed a main peak retention time (RT) of 0.87 min.
[1] Saied M. Soliman, Ahmed M.A. Badr. “Synthesis, structure and Hirshfeld analysis of the potent antimicrobial [Ag(4-bromopyrazole)2ClO4] complex.” Polyhedron 171 (2019): Pages 323-329.
[2] Shaoling Wang , Hongkun Zhao, Cunbin Du . “Solubility, solution thermodynamics and preferential solvation for 4-bromopyrazole in mixed solvents of (methanol/ethanol+ water) from T = (283.15 to 318.15) K.” Journal of Chemical Thermodynamics 112 (2017): Pages 146-154.