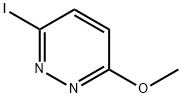
To a solution of 3,6-diiodopyridazine (1.40 g, 4.22 mmol) in methanol (20 mL) was added sodium methanolate (0.34 g, 6.33 mmol) and the reaction mixture was stirred at 80 °C for 1 hour. Upon completion of the reaction, the reaction was quenched by the addition of water (20 mL) and subsequently extracted with dichloromethane (10 mL x 3). The organic layers were combined, dried with anhydrous sodium sulfate, filtered and concentrated under reduced pressure. Finally, purification by silica gel column chromatography afforded 3-iodo-6-methoxypyridazine (0.97 g, 97%) as a colored solid with a melting point of 96-98 °C.
[1] Tetrahedron, 2015, vol. 71, # 40, p. 7670 - 7675
[2] Journal of Medicinal Chemistry, 1999, vol. 42, # 4, p. 669 - 676
[3] Journal of Heterocyclic Chemistry, 1999, vol. 36, # 5, p. 1135 - 1142