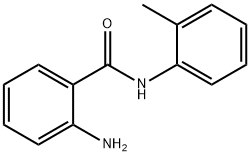
Nitrobenzene (0.6 mmol) was used as a feedstock and mixed with 5 wt% Pd/C (0.5 mol%, 0.003 mmol), H2O (10 eq, 6.0 mmol), B2(OH)4 (3.3 eq, 2.0 mmol) and CH3CN (1.0 mL) in a 10 mL reaction tube. The reaction mixture was stirred at 50 °C for 24 hours. The reaction progress was monitored by TLC. After completion of the reaction, the mixture was cooled to room temperature. Water (5 mL) was added and extracted with EtOAc (3 x 5 mL). The organic phases were combined, dried with anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by silica gel column chromatography to afford the target product 2-amino-N-(o-toluene)benzamide (55 mg, 99% yield).
[1] Tetrahedron, 2017, vol. 73, # 27-28, p. 3898 - 3904
[2] Journal of Medicinal Chemistry, 2008, vol. 51, # 8, p. 2400 - 2411
[3] RSC Advances, 2016, vol. 6, # 33, p. 27378 - 27387
[4] Synlett, 2018, vol. 29, # 13, p. 1765 - 1768
[5] Journal of Medicinal Chemistry, 1986, vol. 29, # 8, p. 1534 - 1537