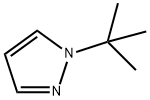
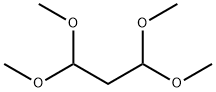
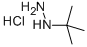GENERAL STEPS: A mixture of 1,1,3,3-tetramethoxypropane (37 g, 226 mmol), tert-butylhydrazine hydrochloride (28 g, 226 mmol) and concentrated hydrochloric acid (60 mL, 720 mmol) in ethanol (300 mL) was placed in a reaction flask. The reaction was heated to reflux overnight. After completion of the reaction, the mixture was poured into water and the aqueous phase was extracted with ether. The organic phases were combined, washed with saturated brine, dried over anhydrous magnesium sulfate and concentrated under reduced pressure to give 1-tert-butyl-1H-pyrazole (25 g, 89% yield). The product was characterized by 1H NMR (400 MHz, DMSO-d6): δ 7.38 (s, 1H), 6.17 (s, 1H), 1.47 (s, 9H); MS (ESI) m/z: 125.1 [M + H]+.