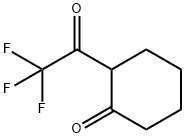
2-(2,2,2-Trifluoroacetyl)cyclohexanone is a useful reactant for the synthesis of fluorinated 1-desazapurines.
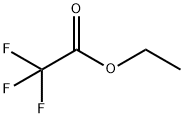
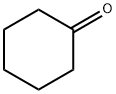
GENERAL PROCEDURE: In a dry Schlenk tube, NaH (6.0 mmol) was stirred with ethyl trifluoroacetate (6.0 mmol) in THF (5 mL) for 10 minutes at room temperature under argon protection. Subsequently, a THF (5 mL) solution of cyclohexanone (5.0 mmol) was added dropwise to the above mixture at 0 °C and under argon protection. The reaction mixture was stirred at the set temperature for 2-6 h. After that, it was cooled to 0 °C again and the reaction was quenched with 6 mL of 1 mol/L HCl solution. After continued stirring for 15 min, the reaction mixture was neutralized with saturated NaHCO3 solution. After conventional post-treatment, the residue was purified by silica gel column chromatography to afford the target product 2-(trifluoroacetyl)cyclohexanone.
[1] Tetrahedron, 2014, vol. 70, # 31, p. 4668 - 4674
[2] Magnetic Resonance in Chemistry, 1993, vol. 31, # 4, p. 323 - 328
[3] Journal of the American Chemical Society, 1953, vol. 75, p. 4753
[4] New Journal of Chemistry, 2007, vol. 31, # 6, p. 936 - 946
[5] RSC Advances, 2016, vol. 6, # 46, p. 40277 - 40286