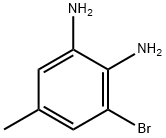
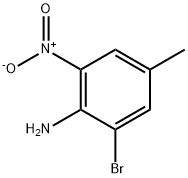
General procedure for the synthesis of 3-bromo-5-toluene-1,2-diamine from 2-bromo-4-methyl-6-nitroaniline: 2-bromo-4-methyl-6-nitroaniline (5.00 g, 21.64 mmol) was dissolved in a solvent mixture of methanol (148 mL) and tetrahydrofuran (18.50 mL). Ammonium chloride (23.15 g, 433 mmol) was added to the solution followed by zinc powder (14.15 g, 216 mmol). The reaction mixture was heated to 40 °C and maintained at this temperature for 1 hour. After completion of the reaction, the mixture was cooled to room temperature and subsequently concentrated under reduced pressure. The concentrate was redissolved in a mixture of ethyl acetate and saturated aqueous sodium carbonate solution with vigorous stirring for 10 minutes. The reaction mixture was filtered through a sintered glass funnel and the solid residue was washed with additional ethyl acetate. The organic layer was separated and washed sequentially twice with water, once with saturated saline and then dried over anhydrous sodium sulfate. The desiccant was removed by filtration and the filtrate was concentrated under reduced pressure to afford the intermediate 3-bromo-5-toluene-1,2-diamine (4.35 g, 21.63 mmol, 100% yield). The product was confirmed by 1H NMR (400 MHz, chloroform-d) and LC-MS (Method H, RT = 0.93 min, MS (ESI) m/z: 201.0 [M + H]+) characterization.