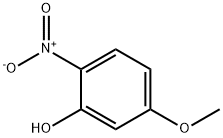
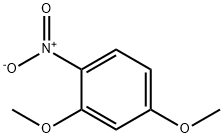
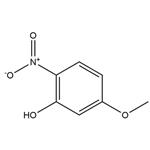
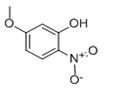
The general procedure for the synthesis of 5-methoxy-2-nitrophenol from 2,4-dimethoxynitrobenzene was as follows: 2,4-dimethoxynitrobenzene (2.0 g, 10.9 mmol) was dissolved in 360 mL of dichloromethane and cooled to 0 °C. Subsequently, anhydrous aluminum trichloride (2.91 g, 21.3 mmol) was added in batches. The reaction mixture was heated to reflux and kept for 5.5 hours (the color of the reaction solution changed to bright red). After completion of the reaction, it was cooled to room temperature and 10% aqueous hydrochloric acid solution was added slowly and dropwise. The mixture was extracted three times with dichloromethane. The organic layers were combined and dried with anhydrous magnesium sulfate. After concentration under reduced pressure to remove the solvent, the crude product was purified by column chromatography (cyclohexane/ethyl acetate = 10:1) to afford 5-methoxy-2-nitrophenol (11) as a yellow solid (1.64 g, 9.69 mmol, 89% yield).