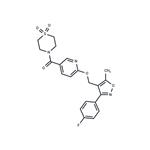
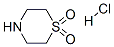1) 6-[3-(4-fluorophenyl)-5-methylisoxazol-4-ylmethoxy]nicotinic acid (23.0 g, 70.1 mmol) and 1,1'-carbonyldiimidazole (15.3 g, 94.6 mmol, 1.35 eq.) were dissolved in THF (120 mL), and the reaction mixture was stirred for 1 hour at room temperature.
2) The above solution was slowly added to a suspension of THF (120 mL) containing thiomorpholine-1,1 -dioxide hydrochloride (16.9 g, 98.5 mmol), DMAP (400 mg, 3.27 mmol) and triethylamine (9.78 g, 96.7 mmol).
3) The reaction mixture was heated to reflux temperature and stirred continuously at this temperature for 50 hours.
4) Upon completion of the reaction, the mixture was cooled to room temperature and then water (300 mL) was slowly added over a period of 1 hour.
5) THF was removed by distillation under reduced pressure at 60 °C, while solvent replacement was carried out by successive addition of ethanol (426 g) in a constant volume.
6) The resulting suspension was cooled to room temperature and stirring was continued for 2 hours.
7) The crystals were collected by filtration and washed with a mixture of ethanol (100 mL) and water (100 mL).
8) Drying to constant weight at 55 °C/<25 mbar afforded 28.9 g (92% yield) of the target product (1,1-dioxothiomorpholine)(6-((3-(4-fluorophenyl)-5-methylisoxazol-4-yl)methoxy)pyridin-3-yl)methanone as a colorless solid with a purity of 99.7% (area) as determined by HPLC.

![3-Pyridinecarboxylic acid, 6-[[3-(4-fluorophenyl)-5-methyl-4-isoxazolyl]methoxy]-](/CAS/20210111/GIF/1159600-32-4.gif)