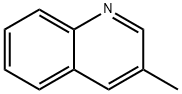
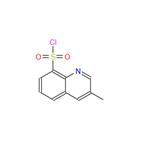
The general procedure for the synthesis of 3-methylquinoline-8-sulfonyl chloride from 3-methylquinoline was as follows: 3-methylquinoline (5.2 g, 36 mmol) was slowly added to 9 mL (135 mmol) of chlorosulfonic acid at 0°C with reference to U.S. Patent Nos. 4,522,576 and 5,332,822. After removing the ice bath, the reaction mixture was stirred at 100 °C overnight. After the reaction mixture was cooled to room temperature, 3.3 mL (45 mmol) of thionyl chloride was added, followed by heating at 70 °C for 1 hour. Upon completion of the reaction, the mixture was cooled to 0 °C and carefully quenched with ice (note: this step is highly reactive). The reaction mixture was diluted with 100 mL of water and extracted with dichloromethane (100 mL). The organic phase was washed with water, dried over anhydrous magnesium sulfate and concentrated to give the crude product. The crude product was ground with a solvent mixture of dichloromethane/ether to give 1.58 g (18% yield) of 3-methylquinoline-8-sulfonyl chloride as a brown solid. The product was characterized by 1H-NMR (300 MHz, DMSO-d6): δ 9.17-9.29 (m, 2H), 8.32-8.38 (m, 2H), 7.96 (dd, 1H, J = 7Hz), and 2.51 (t, 3H, J = 2Hz).