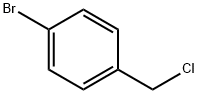
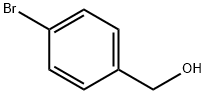
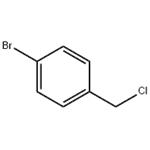
General procedure for the synthesis of 4-bromobenzyl chloride from 4-bromobenzyl alcohol: 4-bromobenzyl alcohol (0.6 g, 0.6 mmol) and dichloromethane (5 mL) were added to a round bottom flask. After stirring for 10 min, oxalyl chloride or oxalyl bromide (0.6 mmol) was slowly added. The reaction mixture was magnetically stirred at room temperature. After the gas was released, 4-bromobenzyl alcohol (0.5 mmol) was added and the reaction mixture was heated to reflux. The progress of the reaction was monitored by TLC and after completion of the reaction, the mixture was cooled to room temperature and filtered. The solid on the funnel was washed with dichloromethane (3 x 10 mL), the filtrate was combined and concentrated under reduced pressure. The purity of the resulting 4-bromobenzyl chloride was confirmed by 1H NMR and 13C NMR spectral analysis.
[1] Beilstein Journal of Organic Chemistry, 2014, vol. 10, p. 1397 - 1405
[2] Organic Letters, 2013, vol. 15, # 1, p. 108 - 111
[3] Organic and Biomolecular Chemistry, 2013, vol. 11, # 24, p. 4016 - 4024
[4] Bulletin of the Korean Chemical Society, 2010, vol. 31, # 11, p. 3434 - 3436
[5] Organic Letters, 2018, vol. 20, # 10, p. 3061 - 3064