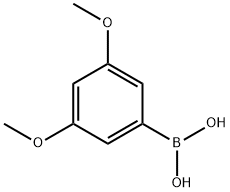
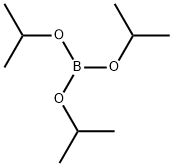
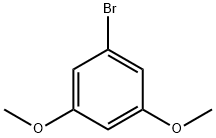
1. Synthesis of 3,5-dimethoxyphenylboronic acid: After cooling the flame-dried reaction vessel under argon protection, 3,5-dimethoxybromobenzene (3 g, 13.82 mmol) and anhydrous tetrahydrofuran (36 ml) were added. The mixture was stirred until a clarified solution was formed. Subsequently, the reaction mixture was cooled to -78 °C and maintained at this temperature for 15 minutes. n-Butyllithium (10.92 ml, 2.1 M hexane solution) was slowly added and the reaction mixture continued to be stirred at -78 °C for 30 minutes. Then, triisopropyl borate (5.2 g, 27.64 mmol) was added dropwise and the reaction was continuously stirred at -78°C for 2 hours. After completion of the reaction, it was slowly warmed up to room temperature and acidified with 2 M sulfuric acid solution to pH 2. The reaction mixture was extracted with ethyl acetate, the organic phases were combined, dried with anhydrous magnesium sulfate and concentrated under reduced pressure to remove the solvent. The crude product was purified by recrystallization from petroleum ether to give 3,5-dimethoxyphenylboronic acid in 92% yield.