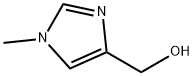
At 0 °C, (1-methyl-1H-imidazol-4-yl)methanol (5 g, 44.64 mmol) was dissolved in dichloromethane (10 mL) and thionyl chloride (50 mL) was added slowly and dropwise. The reaction mixture was stirred overnight at room temperature and then refluxed for 3 hours. Upon completion of the reaction, the solvent was removed by concentration under reduced pressure. The residue was treated with diethyl ether and the resulting precipitate was filtered and dried to give 4-(chloromethyl)-1-methyl-1H-imidazole hydrochloride as a brown solid (4 g, 53.80% yield). The product was characterized by 1H NMR (300 MHz, DMSO-d6): δ 9.25 (s, 1H), 7.8 (s, 1H), 4.95 (s, 2H), 3.9 (s, 3H).