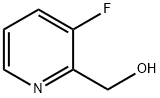
3-Fluoropyridine-2-methanol is a pharmaceutical intermediate used to synthesize compounds with other applications. For example, 3-fluoropyridine-2-methanol can be used as a starting material to synthesize a tetrahydrocarbazole derivative that can be used as an androgen receptor modulator, with the following structure:
Dissolve 215 mg (1.69 mmol) of 3-fluoropyridine-2-methanol in 10 mL of dichloromethane and cool to 0 °C. Add thionyl chloride (160 μL, 2.20 mmol) and stir the reaction mixture for 1 hour. Add 50 mL of dichloromethane, and stir the reaction mixture with 2 × 40 mL of saturated sodium bicarbonate aqueous solution and 2 × 40 mL of brine. The organic fraction was separated and dried over magnesium sulfate, filtered, and concentrated under pressure to give 2-chloromethyl-3-fluoro-pyridine. Sodium hydride (60% in oil, 80 mg, 1.99 mmol) was then suspended in DMF (2 mL) and cooled to 0 °C. A solution of N-(6-cyano-2,3,4,9-tetrahydro-1H-carbazole-3-yl)-isobutyramide (253 mg, 0.90 mmol) in DMF (2 mL) was slowly added via syringe, and the mixture was stirred for 30 minutes, then warmed to ambient temperature for 60 minutes. 2-chloromethyl-5-fluoro-pyridine hydrochloride (180 mg, 0.99 mmol) was added, and the reaction mixture was stirred for approximately 12 hours. The reaction was quenched with a saturated aqueous solution of ammonium chloride (5 mL). Ethyl acetate (50 mL) was added, followed by water (50 mL) and washing with brine (2 × 50 mL). The organic phase was separated, dried over magnesium sulfate, filtered, and evaporated under reduced pressure. The resulting residue was ground with ether (10 mL) for 5 minutes and then filtered to obtain 187 mg (53%) of the above compound as a light yellow solid.